Imagine a single test that could scan a human embryo’s entire genetic blueprint, its whole genome. A test that promises to help prospective parents pick the best and healthiest one, you know, free from a whole laundry list of diseases, together with choosing desirable traits such as high IQ, tallness, and fair complexion.
Well, this is not science fiction anymore. It is an emerging technology that is knocking on our door right now, and it could completely change how we think about having children forever.
So let us just ask a really direct question. If you had the choice, would you do it? Would you pick the best embryo?
It sounds like a no-brainer. Of course, all parents want the best future for their kids! But that one question just cracks open a Pandora’s box of incredibly complicated ethical and moral issues.
At present, a handful of tech companies in the United States like GenEmbryomics, Orchid Health, Juniper Genomics, and Nucleus Genomics now offer such advanced genetic testing, with some of them even marketing embryo testing for IQ.
However, before thinking further, we first need to understand this new revolutionary technology for IVF called PGT-WGS, which stands for Preimplantation Genetic Testing with Whole Genome Sequencing.
Now, here is what makes it such a huge deal. Think of it like this. The older IVF genetic tests we currently have, they were kind of like spot checking a book for a few typos that you already knew about.
In contrast, PGT-WGS is like thoroughly proofreading the entire book at once, looking for everything from tiny spelling mistakes to big plot holes, and is even trying to predict the overall story. It is claimed that this is a massive leap forward in the field of IVF genetic testing.

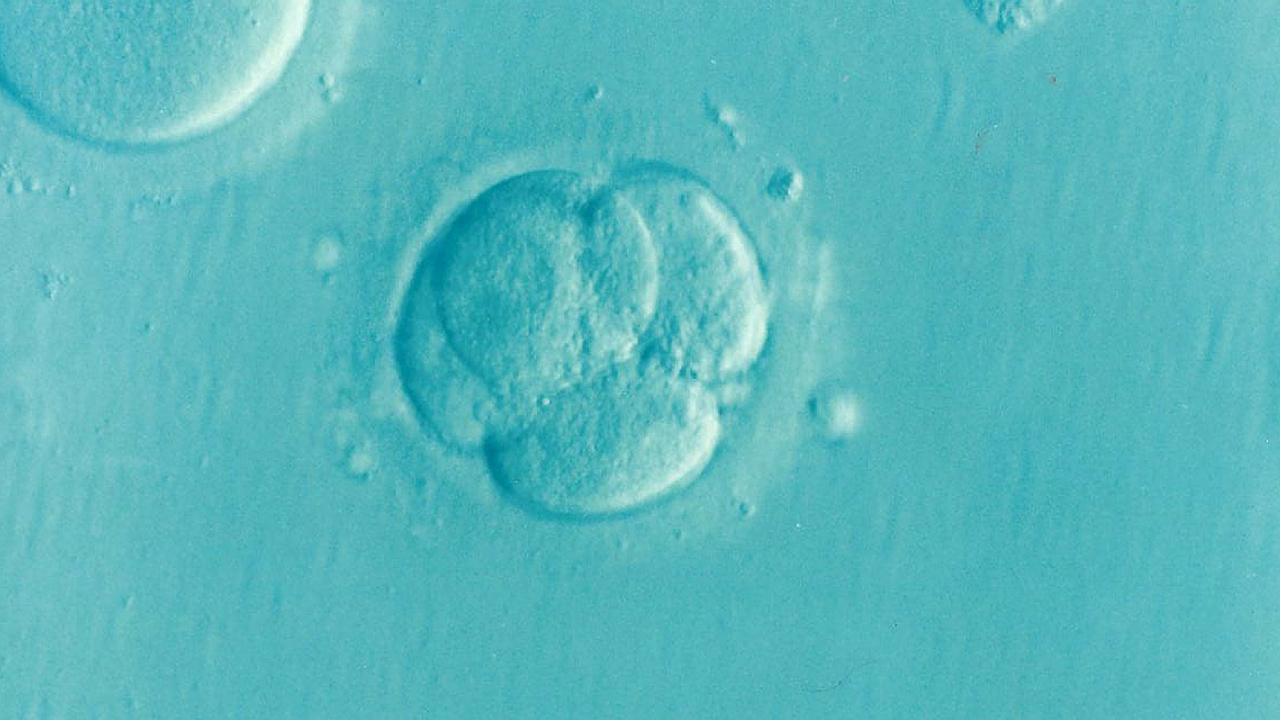
So how does this whole thing work? Well, it all starts with an IVF embryo that is about five days old. It is called a blastocyst.
A tiny sample, just a few cells, is carefully taken from its outer layer, the trophectoderm that goes on to form the placenta and umbilical cord, but not the baby itself.
Then, the DNA from those few cells gets amplified, which means that it is basically photocopied millions and millions of times until there is enough to run a whole genome sequence. And what you get at the end is this massive flood of data covering the embryo’s entire genetic code.
And this is what makes it an absolute game-changer. PGT-WGS basically combines four different tests that used to be separate into one package. You got PGT-A, which checks for big chromosomal problems like the kind that causes Down syndrome.
Then, there is PGT-M, which hunts for single gene disorders like beta thalassemia. It also does PGT-SR, looking for structural mix-ups in the chromosomes.
And then, there is the really controversial one, PGT-P, which calculates risk scores for complex multi-gene disease conditions like type 2 diabetes and maybe for other non-disease traits too, like height and intelligence.
Next, we have to ask what is going on in our society that is creating such a huge demand for a tool like this? Well, there are a few really powerful forces all converging at once.
Global fertility rates are dropping, with more people, especially women, having kids later in life. That means families are often smaller and parents are pouring enormous resources, both financial and emotional, into each child.
And this has helped create what some sociologists call child perfectionism, the phenomenon of heavy-handed tiger parenting and unrealistic parental expectations, which is further exacerbated by an increasingly competitive educational system.
Mix all that with a very real fear about age-related genetic risks in older mothers, such as Down syndrome, and you have created the perfect storm, a huge demand for any technology that promises a healthy, successful child.
Hence, PGT-WGS is not just some neutral medical tool. It plugs directly into our deepest, most modern anxieties and hopes about being a parent. It promises to calm our fears that things might go wrong, while at the same time, it is feeding our desire for that perfect child.
That is a potent mix, and you can bet private fertility clinics and genetic companies are going to market it aggressively, and perhaps unethically as well.
To play on patients’ fears and hopes, unethical “guilt-tripping” marketing tactics might be used, whereby prospective parents are made to feel guilty for not using this powerful technology to give their future children the best start in life.
Social pressure might make it hard to resist using PGT-WGS when you see your circle of friends and relatives using it. Healthy and fertile couples might unnecessarily choose to undergo expensive and invasive IVF so that they can conceive “genetically optimised” kids with PGT-WGS.
Hence, it is imperative to highlight what IVF patients should be aware of before buying into this new “wonder” technology.
First off, the process of extracting cells from the embryos for genetic testing (biopsy procedure) carries a risk of damaging IVF embryos. All previous studies claiming negligible harmful effects of biopsy were based on strong and good quality embryos. Older women tend to produce fewer, weaker, and lower quality embryos that might incur higher risks of damage through biopsy.

Secondly, IVF embryos that appears the healthiest under the microscope, which would likely ensure the best chances of pregnancy may not have the “best genetics” or “lowest genetic risk”, as revealed by PGT-WGS screening.
Hence, some IVF patients may have to consider choosing a “lower-quality embryo” with lesser genetic risk. By doing so, the patient has to trade better chances of pregnancy for lower genetic risks, which could cause significant confusion, anxiety and distress.
Thirdly, the extracted cells for genetic testing come only from the outer embryo layer (trophectoderm) that will become the placenta and umbilical cord, not the baby, and sometimes their genetics do not match.

Fourthly, there is also a risk of misdiagnosis with PGT-WGS. We now know that many embryos flagged as genetically abnormal are actually mosaic, meaning they have a mix of good and bad cells.
And it turns out, many of these can just self-correct and become perfectly healthy babies. So discarding mosaic embryos flagged as abnormal might lower the patient’s chances of pregnancy.


Fifthly, research suggests that IVF embryos can continue to develop new genetic changes in the days between the PGT-A testing and implantation in the womb. Furthermore, the process of Whole Genome Amplification (WGA) required for analysing the small amount of DNA from the biopsy can introduce technical artefacts, which might mask an abnormality, leading to a “false negative” result.
This has led to rare cases whereby transfers of IVF embryos flagged as normal by PGT-A have resulted in pregnancies with chromosomal abnormalities. Because of such risks, most responsible doctors will still recommend standard prenatal screening (such as NIPT or amniocentesis) after PGT-A.
Sixthly, the most common and established part of the PGT-WGS platform, the PGT-A component that screens for chromosomal abnormalities, does not improve IVF success rates. A 2023 analysis of over 133,000 IVF cycles by SART (Society for Assisted Reproductive Technology) found that PGT-A was actually associated with a lower chance of having a baby than just doing IVF without the procedure.
Seventhly, IVF patients must be made aware of what professional bodies in developed countries, such as the US and United Kingdom, are recommending on embryo genetic screening with PGT.
For instance, the American Society for Reproductive Medicine (ASRM) has stated that the broad application of PGT-A on all IVF patients cannot be recommended due to doubtful results on improving live birth rates, and that its effectiveness in lowering the risk of miscarriages also remains unclear.
Likewise, PGT-A has been given a red light rating by the Human Fertilisation and Embryology Authority (HFEA) in the UK, due to doubtful data on its effectiveness in improving IVF success rates.
In neighbouring Singapore, which has strict medical regulations, PGT-A has not yet been approved as a routine clinical procedure, and is still under evaluation by a clinical trial. This is itself a red flag.
Eighthly, IVF patients must be made aware of the current ongoing lawsuits against the PGT-A technique in the US. The plaintiffs allege that they have not been properly informed about the various risks and limitations of the technique, and have been misled into discarding viable embryos, which have robbed them of their chances of parenthood.
In Australia, fertility clinic Monash IVF agreed to compensate A$56 million to over 700 former patients for inaccurate and faulty genetic testing of IVF embryos, which led to the destruction of potentially viable ones.
Ninthly, IVF patients must be aware that the polygenic risk scoring component of PGT-WGS, PGT-P, which evaluates complex multi-gene disease traits like type 2 diabetes, represents just probability rather than certainity of developing specific disease conditions.
These probability measurements are made relative to whole population big data statistics.
Tenthly, IVF patients considering PGT-WGS for selecting complex multi-gene traits such as IQ and height, must be made aware of a genetic phenomenon called “pleiotropy”.
It is basically the idea that one single gene can affect a bunch of different traits, sometimes in ways you would never expect. So you might be selecting an embryo for a few genes you think is great for one thing, but you could be unknowingly signing up for a bunch of other traits that are not so great.
And this is where it gets incredibly real and, frankly, frightening for parents. Some genetic research has suggested that some of the same genes linked to high IQ might also increase the risk for conditions like autism and bipolar disorder.
So what is the choice? Do you roll the dice on a few extra IQ points for a higher risk of a serious neuropsychiatric condition?
Similarly, taller people have higher risks of some diseases, so is it wise to select for height? This is not just a thought experiment. These are the kinds of dizzying choices that this technology could actually force prospective parents to make.
Lastly, patients must be aware that the use of big population genomic data to derive polygenic risk scores for embryo selection may be misguided and inappropriate.
This is due to the limited genetic variability of the few IVF embryos obtained from a single pair of parents, which severely limits the possible outcomes of embryo selection.
Based on computer modelling prediction, even if you have a bunch of embryos to choose from, the average gain you can expect from this kind of screening is about 2.5 cm in height and 2.5 IQ points. Is such a small gain worth all the costs and the risks?
Hence, the arrival of PGT-WGS is way more than just a personal choice for a couple in an IVF clinic. It poses a dilemma for society as a whole, the very real fear of sliding into a new consumer-driven eugenics.
The major concern is that this technology will stop being a medical tool to prevent horrific diseases and start being a cosmetic tool to select for socially desirable traits, things like height, skin colour, and intelligence.
This could end up just reinforcing our worst social biases and encouraging unrealistic expectations that parents have of their genetically optimised kids, after spending do much money on the procedure.
Somewhere, a line has to be drawn between helping a family have a healthy child and creating a new kind of genetic inequality.
This would be the responsibility of the Ministry of Health (MOH), who would have to grapple with permitting and regulating the PGT-WGS procedure.
Dr Alexis Heng Boon Chin, originally from Singapore, is an associate professor of biomedical science at Peking University, China.
- This is the personal opinion of the writer or publication and does not necessarily represent the views of Ova.